 Also look up how isotopes are written (with numbers to the left of the element's symbol). **I would review the definitions for atomic number and mass number. SO, the best answer to your question is "An isotope of Chlorine has 17 protons and 18 neutrons." The average mass of chlorine is the fraction that is 35 Cl times the mass of 35 Cl plus the fraction that is 37 Cl times the mass of. What is the percent composition of Cl in terms of these two isotopes Solution. 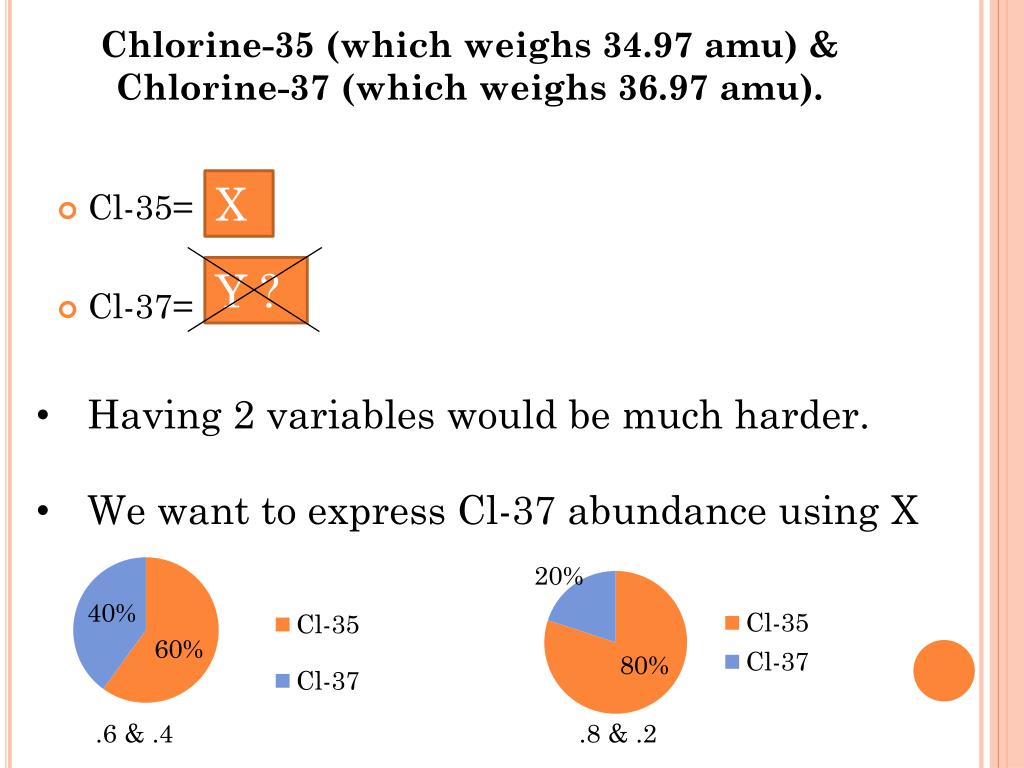
It is an extremely reactive element and a strong oxidising agent: among the elements, it has the highest electron. Chlorine is a yellow-green gas at room temperature. If their exact masses are 34.9689 amu and 36. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine has two stable isotopes, Cl-35 and Cl-37. This means that the average Cl atom has 17 protons and just over 18 neutrons (17 + 18 = 35). Naturally occurring chlorine consists of 35 Cl (mass 34.96885 amu) and 37 Cl (mass 36.96590 amu), with an average mass of 35.453 amu. Chlorine is a chemical element with the symbol Cl and atomic number 17. On the Periodic Table, the average atomic number is listed underneath the Cl symbol as 35.45. We add that decimal to the number of protons that the element ALWAYS has, and end up with the average atomic number. Chlorine-37 accounts for 24.23 of natural chlorine, chlorine-35 accounting for 75.77, giving chlorine atoms in bulk an apparent atomic weight of 35.453 g/mol. (b) 45 protons, 24 neutrons, 43 electrons. Write the nuclide notation, including charge if applicable, for atoms with the following characteristics: (a) 25 protons, 20 neutrons, 24 electrons. Its nucleus contains 17 protons and 20 neutrons for a total of 37 nucleons. For each of the isotopes in Exercise 21.1, determine the numbers of protons, neutrons, and electrons in a neutral atom of the isotope. 
It is impossible to tell how many neutrons a particular atom has, (unless you have some really high tech equipment to work with) so for the sake of chemistry, we sample a bunch of atoms of a given element (Chlorine, in this case) and take the average number of neutrons. Chlorine-37, is one of the stable isotopes of chlorine, the other being chlorine-35. The mass of a regular chlorine atom, Cl (35) is 34.968853 grams (per mole) and the mass of the isotope Cl (37) is 35.965903 grams (per mole). (A Chlorine atom with 17 neutrons would not be considered an isotope, because it has the same number of neutrons as protons) However, ISOTOPES exist, which are atoms with varying numbers of neutrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
